Find resources on reporting serious or continuing noncompliance with the PHS Policy, serious deviations from the provisions of the Guide, and suspensions of animal activities. Here you can find examples of reportable situations, situations where reporting is not normally required, the time frame for reporting, information to be reported, and more.
Get Started
OLAW accepts reports and allegations of noncompliance from institutions and individuals. All of these reports are evaluated by OLAW and substantiated allegations may result in a range of actions by the Office. Read more to learn about noncompliance reports, including what to report, when to report, how OLAW evaluates reports, and potential resulting actions. Although possible, severe compliance actions affecting an award are rare because institutions are usually able to address incidents successfully and take appropriate actions to prevent reoccurrence.
Policies and Laws
Health Research Extension Act of 1985 (Public Law 99-158)
"(d) If the Director of NIH determines that...
- the conditions of animal care, treatment, or use in an entity which is receiving a grant, contract, or cooperative agreement involving research on animals under this title do not meet applicable guidelines established under subsection (a);
- the entity has been notified by the Director of NIH of such determination and has been given a reasonable opportunity to take corrective action; and
- no action has been taken by the entity to correct such conditions;
the Director of NIH shall suspend or revoke such grant or contract under such conditions as the Director determines appropriate."
- "The IACUC, through the Institutional Official, shall promptly provide OLAW with a full explanation of the circumstances and actions taken with respect to:
- any serious or continuing noncompliance with this Policy;
- any serious deviation from the provisions of the Guide;
- any suspension of an activity by the IACUC."
- "The IACUC may suspend an activity which it previously approved if it determines that the activity is not being conducted in accordance with applicable provisions of the Animal Welfare Act, the Guide, the institution's Assurance, or IV.C.1.a.-g. of [the PHS] Policy. The IACUC may suspend an activity only after review of the matter at a convened meeting of a quorum of the IACUC and with the suspension vote of a majority of the quorum present."
- "If the IACUC suspends an activity involving animals, the Institutional Official in consultation with the IACUC shall review the reasons for suspension, take appropriate corrective action, and report that action with a full explanation to OLAW."
Guidance
Guidance on Prompt Reporting to OLAW Under the PHS Policy on Humane Care and Use of Laboratory Animals
| Notice | Description | Date |
|---|---|---|
| NOT-OD-25-148 | Notice on Update to Guidance on Prompt Reporting to OLAW Under the PHS Policy on Humane Care and Use of Laboratory Animals (replaces the rescinded Notice NOT-OD-05-034) | January 20, 2026 |
| NOT-OD-13-044 | Notice of Change to Electronic Submission of Final Noncompliance Reports to the Office of Laboratory Animal Welfare | February 21, 2013 |
| NOT-OD-10-081 | Guidance on Confirming Appropriate Charges to NIH Awards During Periods of Noncompliance for Activities Involving Animals | April 15, 2010 |
| NOT-OD-07-044 | NIH Policy on Allowable Costs for Grant Activities Involving Animals When Terms and Conditions are not Upheld | January 26, 2007 |
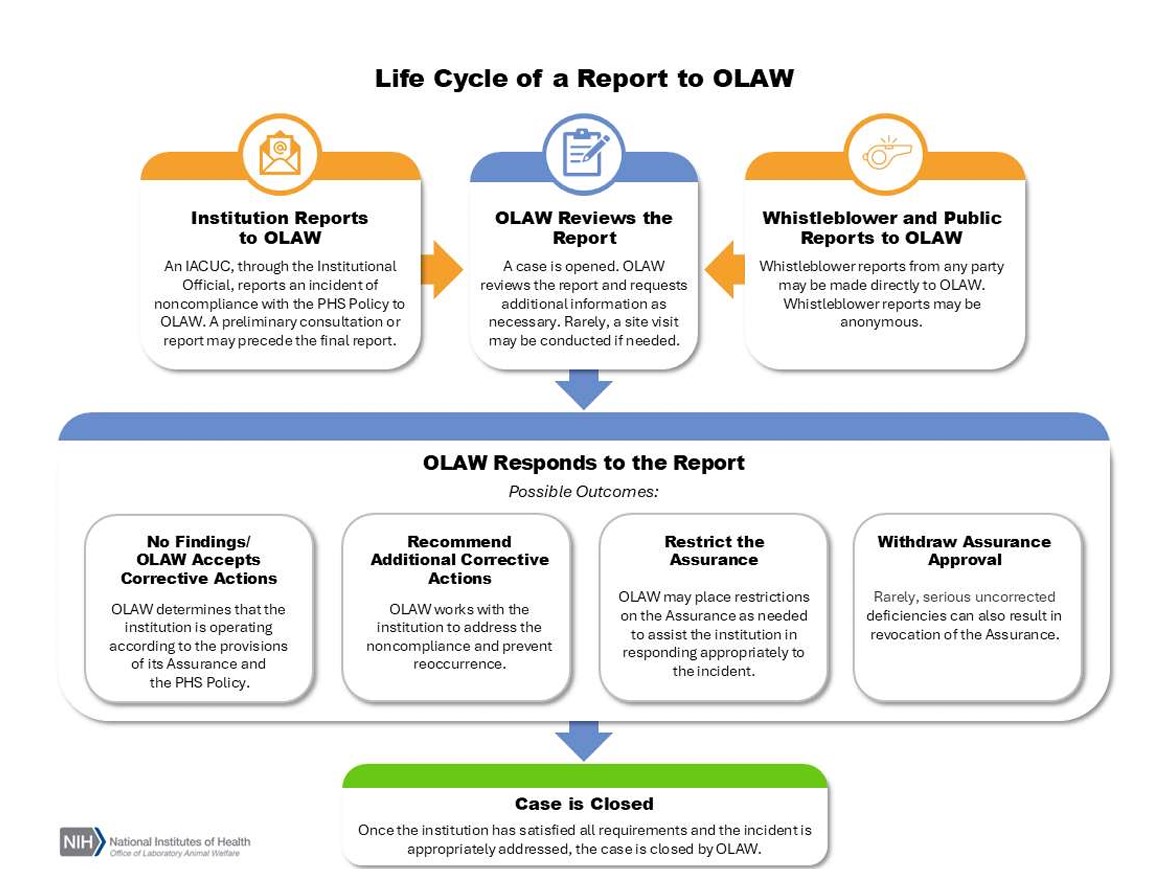
Life Cycle of a Report to OLAW
-
OLAW Receives a Report
Reports arrive at OLAW's Division of Compliance Oversight in one of two ways: either they are submitted by an institution as a self-report, or they are directly communicated to OLAW by a third party. (For guidance on institutional responsibilities regarding whistleblowers and whistleblower protections, see FAQ. G.14.) If submitted by an institution, information should be communicated as a preliminary report via phone or email as soon as possible, even if an investigation is ongoing. This allows OLAW to provide assistance throughout the investigation and corrective process, especially if it is expected to be protracted. The final report can be submitted once additional information has been gathered and corrective actions have been determined.
-
OLAW Reviews and Responds to the Report
OLAW reviews all reports and allegations of noncompliance. Generally, questions can be resolved through dialogue between OLAW and the institution. If a whistleblower or public report is received, institutions will be notified of the nature of the complaint and, except in rare circumstances, have the opportunity to undertake corrective actions. If OLAW is unable to substantiate the allegations in the report after review and a request for additional information, or if the case is substantiated and the institution has implemented appropriate corrective actions, the case is closed by OLAW. If additional straightforward corrective actions are necessary before the case can be closed, OLAW works with the institution to identify and implement any remaining steps to address the noncompliance and prevent recurrence.
Though uncommon, OLAW may take additional action to assist the institution in responding appropriately to the incident, such as:
- Placing the institution on an enhanced reporting schedule, where regular follow-up reports are required to demonstrate continued effectiveness of corrective actions;
- Performing further in-depth review via a site visit;
- Restricting the Assurance applicability for some activities;
- Requiring OLAW review of all Assured activities, etc.
Rarely, serious uncorrected deficiencies can also result in revocation of the Assurance. However, compliance actions affecting an award are rare because institutions are usually able to address incidents successfully and take appropriate actions to prevent reoccurrence.
-
The Case is Closed
Once the institution has satisfied all requirements and the incident is appropriately addressed, the case is closed by OLAW.
For additional information regarding OLAW's process of evaluation, including a detailed explanation of the sequence of events and outcomes of OLAW's review, see the memo "Compliance Oversight Procedures."
A flowchart of the compliance review process can be accessed by clicking the thumbnail. The expanded image will open in your browser.
Download PDF Version of Flowchart (PDF, 93.4 KB)
Examples Based on Reporting Category
Institutions are reminded that the requirements for reporting extend to all live, vertebrate animal activities covered by the PHS Policy. Those institutions whose Assurance states that all incidents will be reported as required regardless of funding source must report accordingly. While the IACUC has the duty to report based on consideration of the circumstances, OLAW reserves the right to make the final determination regarding the reportability of an event. For this reason, a preliminary call to OLAW is always encouraged if there are questions about reporting.
Situations that meet the provisions of section IV.F.3 and are identified by external entities such as the United States Department of Agriculture (USDA) or AAALAC International, or by individuals outside the IACUC or outside the institution, are not exempt from reporting.
Examples When Reporting is Required
As a comprehensive list of definitive examples of reportable situations is impractical, the examples below do not cover all instances but demonstrate the threshold at which OLAW expects to receive a report. Items not listed may still qualify for reporting. Institutions should use rational judgment in determining what situations meet the provisions of PHS Policy section IV.F.3. and fall within the scope of the examples below and consult with OLAW if in doubt. OLAW welcomes inquiries and discussion and will provide guidance regarding specific situations.
The following text is taken from NOT-OD-25-148:
| IACUC Function and Protocol Review | Animal Management | Program Management |
|---|---|---|
|
|
|
Examples of Situations Where Reporting is Not Normally Required
OLAW recognizes that there may be levels of illness and death in virtually any animal-related activity. Accordingly, there may be levels of morbidity and mortality associated with the care and use of animals in research, research training, experimentation, or biological testing or for related purposes that are not the result of violations of the PHS Policy or serious deviation from provisions of the Guide. The examples below typically do not meet the threshold for reporting; however, the IACUC is to consider the specific circumstances, intent, and actual effect to animal welfare before electing not to report. The following text is taken from NOT-OD-25-148:
IACUC Function and Protocol Review | Animal Management | Program Management |
|---|---|---|
|
|
|
Submitting Noncompliance Reports
Preliminary reports may be made via phone or email to the Division of Compliance Oversight. All final institutional self-reports should be sent via email, through the IO, in PDF format (see NOT-OD-13-044). The signature of the IO provides verification that this requirement is met. Use the dropdowns below to identify what and when to report.
Preliminary self-reports provide a flexibility for institutions to meet the requirement for prompt reporting even though it may take time to fully investigate reportable circumstances and implement appropriate corrective actions. In addition, this flexibility allows OLAW to work with institutions during the investigative process to determine if the circumstances meet the requirements for reporting and provide guidance prior to receipt of a formal written report. Include as many of the following items as possible in the preliminary report to OLAW (the following list taken from NOT-OD-13-044):
- Assurance number
- Award numbers for National Science Foundation (NSF) activities. Only NSF award numbers must be included in the report. Other award numbers need not be included unless requested by OLAW or if determined to be applicable by the reporting institution.
- Funding source for all PHS supported activities, including those having an MOU with OLAW. E.g., NIH, NSF, National Aeronautics and Space Administration (NASA), U.S. Department of Veterans Affairs (VA)
- Complete explanation of the situation, including what happened, when and where, species of animal(s) involved, and category of individual(s) involved (e.g., Principal Investigator or Co-Principal Investigator, technician, animal caretaker, student, veterinarian, etc.)
- A full description of any situation, that in the judgment of the IACUC and Institutional Official, may be a threat to PHS-supported activities
- Description of actions taken by the institution to address the situation
- Description of short- or long-term corrective plans and implementation schedule(s)
The final report should identify the date of the preliminary report, address any of the above not included in the preliminary report, and provide any updates or changes since the preliminary report, if made.

[YOUR INSTITUTION’S NAME AND ADDRESS]
[DATE]
Director, Division of Compliance Oversight
Office of Laboratory Animal Welfare
National Institutes of Health
Dear OLAW:
[YOUR INSTITUTION’S NAME], in accordance with Assurance [ASSURANCE NUMBER] and PHS Policy IV.F.3., provides this report of noncompliance regarding an incident to induce hibernation in ground squirrels, an activity not reviewed and approved by the IACUC in a Principal Investigator’s (PI) animal study protocol, entitled “Metabolism of polyphenols in ground squirrels.” This incident was first reported to OLAW on [DATE] via a telephone call by Justin Thyme, DVM, DACLAM, Associate Director, Department of Medical Pharmacology.
On [DATE], the Office of Research Compliance received information that pilot experiments to induce hibernation in ground squirrels were performed without prior IACUC review and approval. Some animals died during this pilot experiment. At the time of the discovery of the incident, the protocol did not describe procedures for inducing and maintaining hibernation. The IACUC discussed the incident and requested corrective actions from the PI. The activity was funded by an NIH grant.
To address the situation, corrective actions were taken including the submission of an Animal Study Protocol Amendment to request a procedural change describing the induction and maintenance of hibernation in ground squirrels. Approval to procure additional ground squirrels was withheld until the IACUC reviewed and approved the PI’s response, the corrective action plan and protocol amendment for the procedural change.
The PI expressed in a letter to the IACUC that this was an unfortunate incident and took responsibility for the noncompliance. The PI assured the IACUC that an incident of such nature would not occur again. The IACUC determined that this was an isolated incident and not a programmatic failure.
The following corrective action plan was implemented after the incident:
- The PI submitted correspondence to the IACUC expressing regret for the noncompliance incident and that exclusion of the hibernation procedure from the protocol was an oversight.
- The PI described corrective measures that had been implemented to prevent recurrence of an incident of this nature. The measures included informing all research staff of the IACUC’s requirement for securing approval for all procedures prior to work involving animals.
- The PI assured the IACUC that all research staff will from now on carefully review and adhere to approved study protocols.
- The PI submitted a request for a procedural change (Animal Study Protocol Amendment) to the protocol. The amendment describing the procedure to perform a pilot experiment to induce and maintain hibernation in ground squirrels was approved by the IACUC on [DATE].
At their meeting on [DATE], the IACUC also reviewed and accepted the corrective action plan as submitted by the PI. [INSTITUTUION] is committed to protecting the welfare of animals used in research and appreciates the guidance and assistance provided by OLAW in this regard. Should you have any questions regarding this report, please contact [INSTUTIONAL CONTACT].
Thank you for your consideration of this matter.
Sincerely,
[INSTITUTIONAL OFFICAL NAME & CREDENTIALS]
In addition to self-reports from institutions, OLAW accepts concerns from other interested parties, including whistleblower and anonymous reports. Whistleblower and anonymous reports should be submitted by phone or email to the Division of Compliance Oversight at [email protected]. Although OLAW will accept a whistleblower's report, OLAW encourages all parties to first consider using institutional reporting processes for animal welfare concerns. OLAW may withhold identifying information to protect whistleblowers; however, individuals should be aware that they may still be identified by others due to the particular circumstances surrounding the report. Protection from reprisal for whistleblowers must be addressed at the institutional level and according to applicable law. OLAW cannot guarantee that individuals making a report will be protected from adverse institutional actions. See also FAQ. G.14
FOIA
Institutions and individuals are reminded that case documents submitted to OLAW, including email communications and preliminary reports, may be publicly available under the Freedom of Information Act (FOIA) once cases are closed. To learn more about the NIH FOIA process, see FAQ C.4..
FAQs
- What kinds of situations should be reported to OLAW under IV.F.3. of the PHS Policy, and when, where, and how should they be reported?
- Should the IACUC report sanctions other than suspensions that are imposed by the Committee or by other institutional officials?
- Are all documents submitted to OLAW subject to the Freedom of Information Act?
- Are institutions required by FOIA to release information about their research, animal care programs, and IACUCs?
- What are PHS Policy reporting requirements for departures from the Guide?
- What are the institution's responsibilities regarding whistleblowers?