The 21st Century Cures Act was signed into law on December 13, 2016. It’s purpose was to accelerate research to improve human health, and to that end, it contained the mandate that federal agencies reduce the administrative burden on investigators.
About the 21st Century Cures Act
The 21st Century Cures Act (PDF, 929 KB) was signed into law on December 13, 2016. It’s purpose was to accelerate research to improve human health, and to that end, it contained the mandate that federal agencies reduce the administrative burden on investigators.
Regarding research with animals, Section 2034(d) of the law mandated that the NIH, USDA, and FDA review policies and procedures and identify steps to reduce administrative burden on investigators. It also mandated that any actions in response to the 21st Century Cures Act must maintain the integrity and credibility of research findings and the protection of research animals.
Working Group
The NIH, FDA, and USDA convened a Working Group of federal subject matter experts that carried out a review and prepared a draft report of its recommendations as directed by the 21st Century Cures Act. To identify inconsistent, overlapping, and unnecessarily duplicative regulations and policies, the Working Group:
- reviewed published reports, communications, and surveys highlighting the regulations and policies that contribute to investigators' administrative burden;
- conducted listening sessions and met with organizations and stakeholders; and
- issued a Request for Information on possible actions and analyzed stakeholder responses.
These efforts are described in more detail below.
Following this research and analysis, the Working Group sought input through a Request for Information on the draft report and proposed recommendations to reduce the administrative burden associated with research activities with laboratory animals.
The Working Group reviewed numerous reports and surveys that have been published over the last several years addressing the topic of administrative and investigator burden.
The following documents were reviewed:
- Rebuttal to Federation of American Societies for Experimental Biology’s Reforming Animal Research Regulations (PDF, 395.52 KB) 2018, People for the Ethical Treatment of Animals.
- Animal Welfare Regulations Must Not Be Compromised to Comply with the Goals of the 21st Century Cures Act (PDF, 9.67 MB) 2018, Humane Society of the United States and Humane Society Legislative Fund.
- Reforming Animal Research Regulations: Workshop Recommendations to Reduce Regulatory Burden (PDF, 812.26 KB) 2017, Report of an April 17, 2017 workshop organized by Federation of American Societies for Experimental Biology, Association of American Medical Colleges, and the Council on Governmental Relations, with assistance from National Association for Biomedical Research.
- Revising the Requirements for Prompt Reporting under PHS Policy IV.F.3. (PDF, 107.27 KB) 2017, National Association for Biomedical Research.
- Optimizing the Nation’s Investment in Academic Research: A New Regulatory Framework for the 21st Century 2016, National Academies.
- Reducing Investigators’ Administrative Workload for Federally Funded Research (PDF, 2.16 MB ) 2014, National Science Board, National Science Foundation.
- 2012 Faculty Workload Survey Research Report (PDF, 2.1 MB) 2014, Federal Demonstration Partnership.
- Findings of the FASEB Survey on Administrative Burden (PDF, 2.1 MB) 2013, Federation of American Societies for Experimental Biology.
The Working Group conducted listening sessions and met with organizations and stakeholders to obtain their individual perspectives.
Listening Session: January 9, 2018
Representatives from the NIH OLAW, USDA APHIS AC, and FDA held a public listening session at the Hyatt Regency Capitol Hill, Washington, DC, immediately following the Federal Demonstration Partnership (FDP) sessions on January 9th. Provided here are the 01/09/2018 introductory slides (PDF, 166.76 KB) and a list of registered speakers with their written comments if provided.
Speakers and Comments
- Ellen Paul, Ornithological Council – Comments (PDF, 29.63 KB)
- Norman Peterson, MedImmune
- Sue Leary, Alternatives Research & Development Foundation and the American Anti-Vivisection Society
- Alka Chandna, People for the Ethical Treatment of Animals – Comments (PDF, 31.26 KB)
- Jennifer Ball, Humane Society of the United States – Comments (PDF, 46.30 KB)
- Cathy Liss, Animal Welfare Institute – Comments (PDF, 118.12 KB)
- Ryan Merkley, Physicians Committee for Responsible Medicine – Comments (PDF, 109.48 KB)
- J.R. Haywood, Michigan State University – Comments (PDF, 50.29 KB)
- Molly Greene, Michigan State University – Comments (PDF, 112.07 KB)
- Stuart Leland, Princeton University and American College of Laboratory Animal Medicine – Comments (PDF, 57.59 KB)
Listening Session: January 29, 2018
Representatives from the NIH OLAW, USDA APHIS AC, and FDA joined the AAALAC International Council Meeting for a listening session on January 29th. Provided here are the 01/29/18 introductory slides (PDF, 157.97 KB) and a 01/29/18 summary of the comments (PDF, 99.60 KB).
Q&A Session: March 12, 2018
Representatives from the NIH OLAW, USDA APHIS AC, and FDA met with the Animal Welfare Institute, Humane Society Legislative Fund, Physicians Committee for Responsible Medicine, and Humane Society of the United States for a Q&A session on March 12th. Provided here are the 03/12/18 meeting notes (PDF, 152.15 KB).
21st Century Cures Act Initiatives
Note
Initiative | Overview | What We’ve Done... |
|---|---|---|
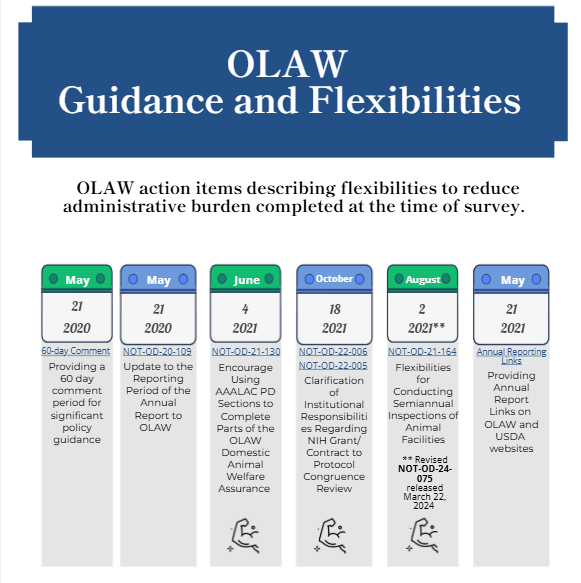
60 Day Comment Period | The NIH Office of Laboratory Animal Welfare (OLAW) committed to providing a minimum of 60 days for comments to significant policy guidance. | The 60-day comment period includes,
Such guidance will focus on high-risk animal welfare concerns affecting institutions, IACUC functions, and updates to guidance as an outcome of the 21st Century Cures Act. |
| AAALAC Program Description | OLAW committed to encouraging AAALAC-accredited institutions to use sections of the AAALAC International (AAALAC) Program Description (PD) in applicable parts of the OLAW Animal Welfare Assurance (Assurance). | OLAW provided information on using sections of the AAALAC International AAALAC PD) to complete parts of the OLAW Domestic Animal Welfare Assurance. Institutions must ensure that all information requested in the Assurance document is provided, including information that is not requested in the AAALAC PD but is required by OLAW. See NOT-OD-21-130 |
Animal Activities Exempt from IACUC Review Under the PHS Policy | OLAW committed to clarifying animal activities that are exempt from the Public Health Service Policy on Humane Care and Use of Laboratory Animals (PHS Policy) requirements for Institutional Animal Care and Use Committee (IACUC) review. | The NIH Office of Laboratory Animal Welfare (OLAW) proposed guidance clarifying animal activities that are exempt from the PHS Policy requirements for IACUC review. Comments were accepted through a Request for Information (RFI) between April 25 and July 31, 2023. |
| Annual Report to OLAW | The 21st Century Cures Act Working Group identified harmonizing the OLAW and USDA annual reporting schedules as an opportunity to decrease administrative burden. | The reporting period for the Annual Report to OLAW has been harmonized with that of USDA. The reporting period is now October 1 – September 30 of each year and must be submitted to OLAW by December 1. See NOT-OD-20-109 |
| Departures from the Guide | The public identified departures from the Guide for the Care and Use of Laboratory Animals (Guide) as an area to reduce administrative burden, and OLAW committed to clarifying the guidance for the reporting requirements of departures from the Guide. | The NIH Office of Laboratory Animal Welfare (OLAW) proposed updates to NOT-OD-12-148 for Departures from the Guide through a Request for Information (RFI) between July 20 and November 1, 2021. |
| Grant and Contract to Protocol Congruence Review | OLAW clarified the requirements, responsibility, timing, and conduct of grant and contract to protocol congruency review for NIH grant applications and contract proposals involving research with live vertebrate animals in response to the 21st CCA. | Clarifications are described in these notices: See NOT-OD-22-005 and NOT-OD-22-006 |
| OLAW Guidance Disclaimer | OLAW committed to clarifying its guidance disclaimer. | Clarifications are described in these notice: See also NOT-OD-25-145 |
| Reporting Noncompliance | OLAW committed to providing clarification on noncompliance reporting. | OLAW provided updated guidance with additional examples of reportable situations, situations where reporting is not normally required, the time frame for reporting, and the information to be reported. See NOT-OD-25-148 |
| Semiannual Animal Facility Inspection | OLAW and USDA committed to clarifying existing flexibilities for conducting semiannual facility inspections. | The Agencies clarified existing flexibilities while maintaining protection for research animals and data integrity. See NOT-OD-24-075 |
| Semiannual Program Review | OLAW and USDA committed to clarifying flexibilities for conducting semiannual program reviews as an opportunity to reduce administrative burden. | The Agencies clarified flexibilities to reduce burden while maintaining protection for research animals and data integrity. See NOT-OD-24-076 |
| Streamlining Protocol Review | OLAW committed to reviewing and revising the current guidance on streamlining the IACUC protocol review process for animal activities. | The NIH Office of Laboratory Animal Welfare (OLAW) proposed updates to NOT-OD-09-035 between July 11, 2023 and October 11, 2023. See NOT-OD-23-152 The comment period is now closed. The final guidance will be published along with a new webpage once comments have been considered. Until comments have been considered and new guidance has been finalized, OLAW expects institutions to comply with applicable existing guidance. |
| Zebrafish | One action identified by the research community was to consider changing the applicability of the PHS Policy to zebrafish larvae from immediately after hatching (typically 3 days post fertilization [dpf] under optimal conditions) to when larvae begin free feeding (at approximately 5 – 7 dpf). OLAW committed to review existing guidance, clarify the requirements, and seek public comment on updated guidance. | While OLAW encourages institutions to use flexibilities to reduce administrative burden while using zebrafish in research), the PHS Policy continues to cover zebrafish larvae immediately after hatching. OLAW sought input on flexibilities to reduce administrative burden while continuing to apply the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals (Policy) to zebrafish (Danio rerio) immediately after hatching in a request for information between May 7, 2021 and August 9, 2021. See NOT-OD 21-118 The comment period is now closed. Additional information on flexibilities will be provided once comments have been considered. |
Policies and Laws
Public Law 114-255 Section 2034(d) (PDF, 929.25 KB) ANIMAL CARE AND USE IN RESEARCH - Not later than 2 years after the date of enactment of this Act, the Director of National Institutes of Health, in collaboration with the Secretary of Agriculture and the Commissioner of Food and Drugs, shall complete a review of applicable regulations and policies for the care and use of laboratory animals and make revisions, as appropriate, to reduce administrative burden on investigators while maintaining the integrity and credibility of research findings and protection of research animals. In carrying out this effort, the Director of the National Institutes of Health shall seek the input of experts, as appropriate. The Director of the National Institutes of Health shall -
- identify ways to ensure such regulations and policies are not inconsistent, overlapping, or unnecessarily duplicative, including with respect to inspection and review requirements by Federal agencies and accrediting associations;
- take steps to eliminate or reduce identified inconsistencies, overlap, or duplication among such regulations and policies; and
- take other actions, as appropriate, to improve the coordination of regulations and policies with respect to research with laboratory animals.
Federal Register Notice 2019-18611 - Laboratory Animal Welfare: Report on Reducing Administrative Burden for Researchers: Animal Care and Use in Research
Federal Register Notice 2018-26557 - Laboratory Animal Welfare: Draft Report on Recommendations to Reduce Administrative Burden on Researchers.
Federal Register Notice 2021-25614 - AWA Research Facility Registration Updates, Reviews, and Reports
Get Started
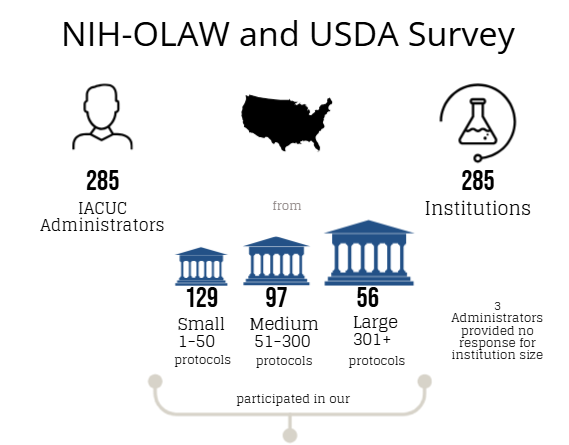
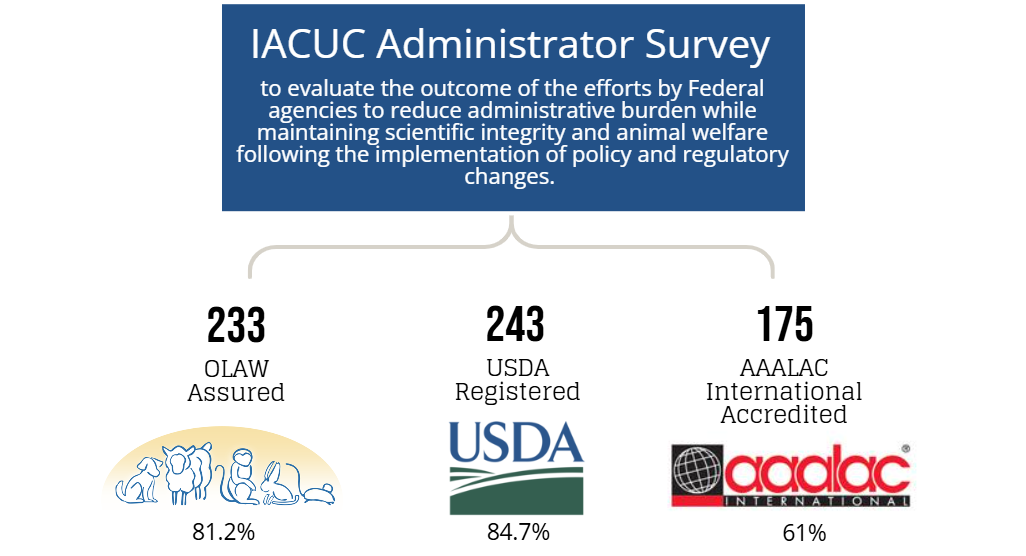
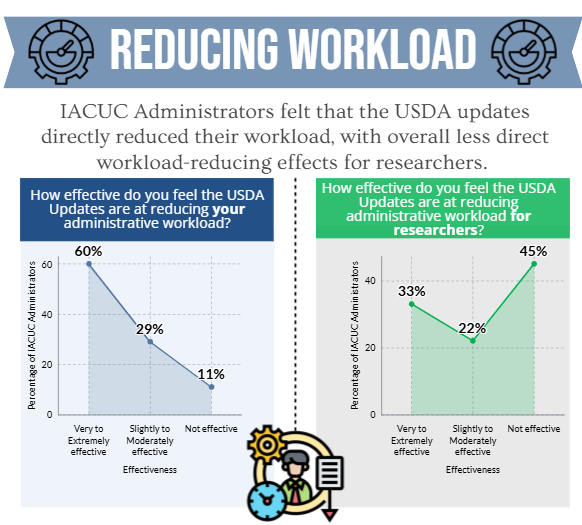
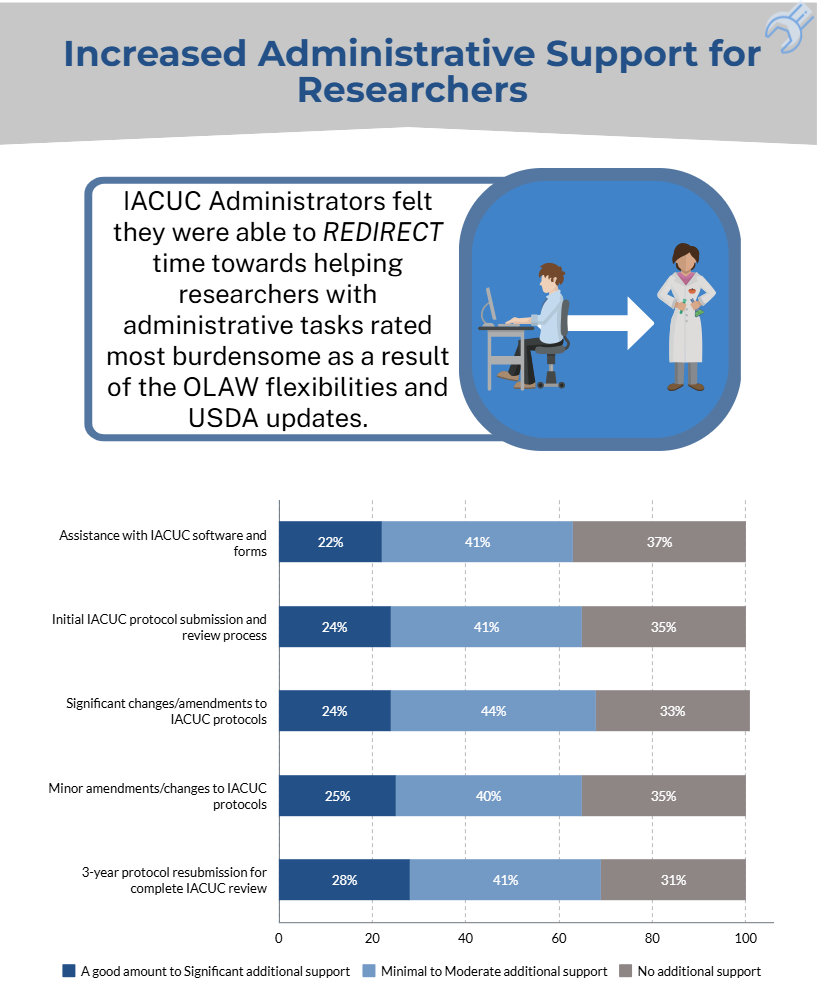
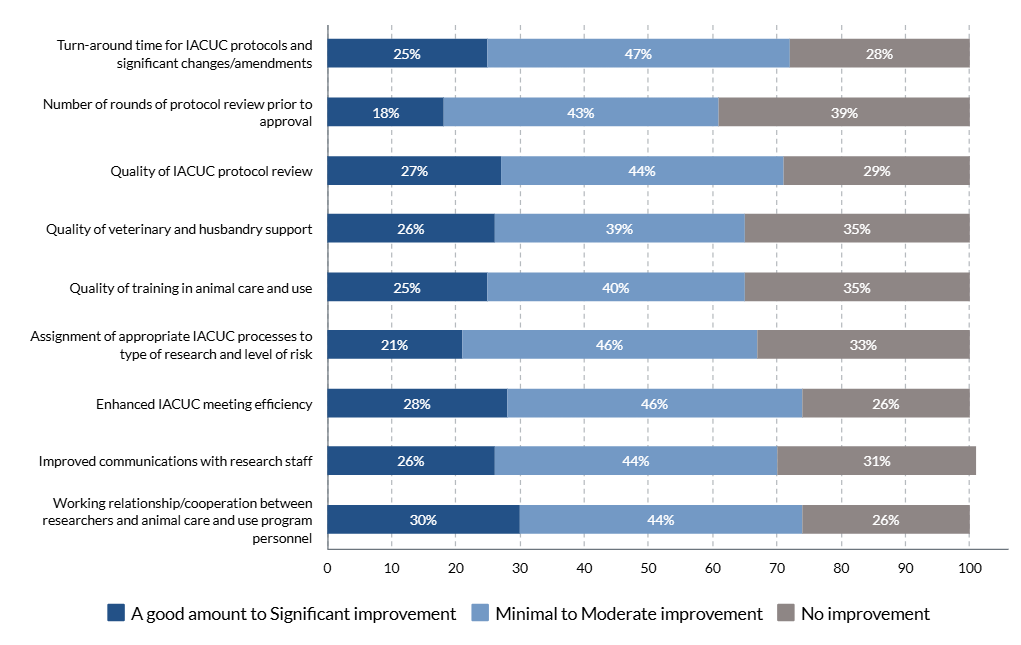
An online Qualtrics survey was distributed from June 1-September 15, 2023, to individuals (e.g., IACUC administrators, coordinators, etc.) whose responsibilities include a direct role in the oversight of their IACUC program and/or IACUC policies and operations at approximately 1,470 OLAW Domestic Assured and USDA-Registered institutions or facilities. The objective of this survey was to determine if:
- institutions implemented optional OLAW flexibilities in their animal care and use programs and
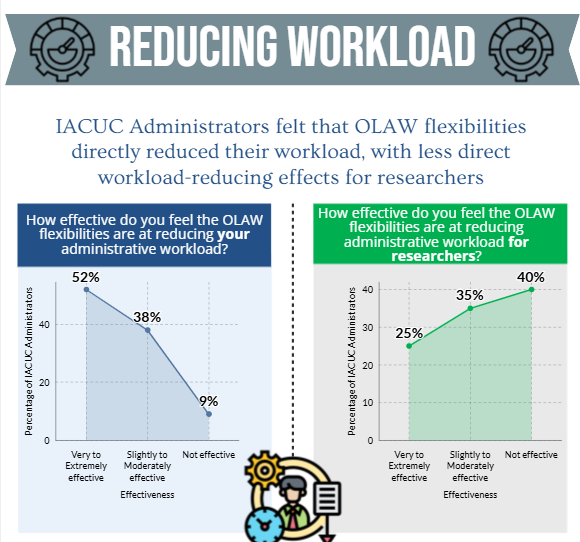
- the OLAW flexibilities and USDA updates directly and/or indirectly reduced administrative workload for administrative staff and investigators.
The results of the IACUC Administrator surveys will help to inform the development of a survey geared towards investigators.
Quick References: Results from the IACUC Administrator Survey